|
This is when he noticed certain types of elements regularly appearing and noticed a correlation between atomic weight and chemical properties.īut the exact Eureka! moment that led Mendeleev to the sorting strategy that produced his complete periodic table is shrouded in mystery. So according to the Royal Society of Chemistry, Mendeleev wrote the properties of each element on cards, and then he started ordering them by increasing atomic weight. But they weren't enough to usefully sort the 55 additional chemical elements known at the time. Gordin in his book "A Well-Ordered Thing: Dmitrii Mendeleev and the Shadow of the Periodic Table" (Princeton University Press, Revised Edition 2018). The first section of Mendeleev's book dealt with just eight of the known elements - carbon, hydrogen, oxygen, nitrogen, chlorine, fluorine, bromine and iodine - and those two strategies worked for those particular elements, according to Michael D. Just two strategies existed at the time to categorize these elements: separating them into metals and nonmetals or grouping them by an element's number of valence electrons (or those electrons in the outermost shell). At the time, there were 63 known chemical elements, each with an atomic weight calculated using Avogadro's hypothesis, which states that equal volumes of gases, when kept at the same temperature and pressure, hold the same number of molecules. Putting the elements in any kind of order would prove quite difficult. (Image credit: Oxford Science Archive/Print Collector/Getty Images) For instance, all the group 18 elements are inert gases, meaning they don't react with any other elements. 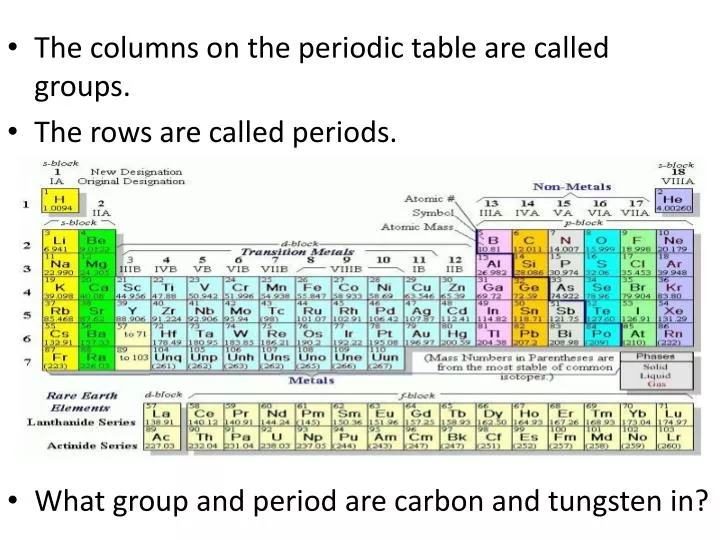
Elements that occupy the same column on the periodic table (called a "group") have identical valence electron configurations and consequently behave in a similar fashion chemically. As an example, elements in Group 8A (or VIIIA) all have a full set of eight electrons in the highest-energy orbital, according to chemist William Reusch, on his webpage at Michigan State University. The columns, or groups, on the periodic table represent the atomic elements that have the same number of valence electrons, or those electrons in the outermost orbital shell. (Atoms have protons and neutrons in their nucleus, and surrounding that, they have their electrons arranged in orbitals, where an atomic orbital is a math term that describes the location of an electron as well as its wave-like behavior.)įor instance, period 1 includes elements that have one atomic orbital where electrons spin period 2 has two atomic orbitals, period 3 has three and so on up to period 7. Unfortunately, there was a slightly different system in place in Europe.The horizontal rows on the periodic table are called periods, where each period number indicates the number of orbitals for the elements in that row, according to Los Alamos National Laboratory. The first two groups are 1A and 2A, while the last six groups are 3A through 8A. The traditional system used in the United States involves the use of the letters A and B. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. 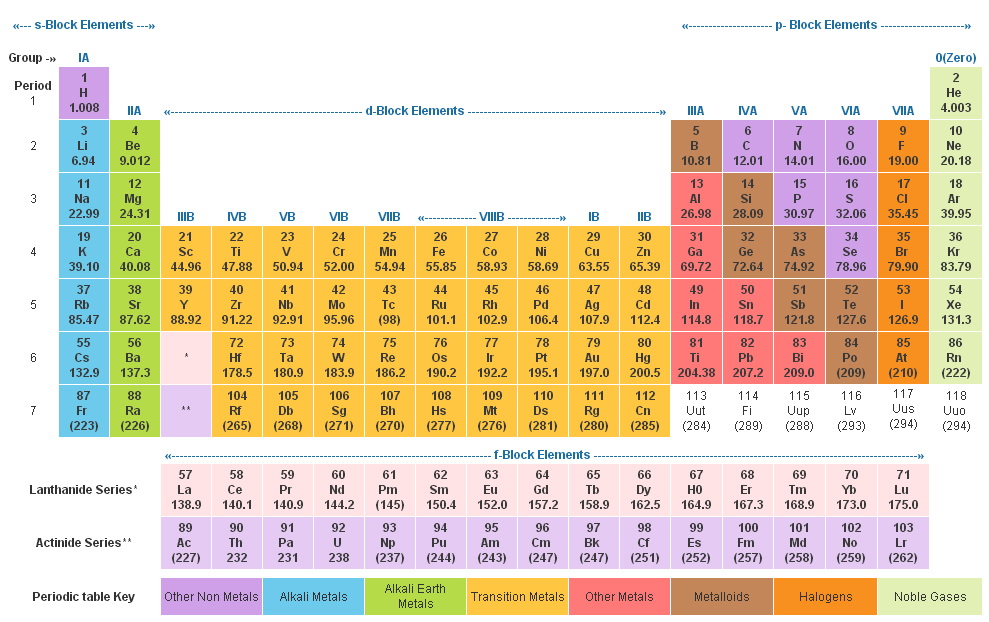
A new period begins when a new principal energy level begins filling with electrons. 
There are seven periods in the periodic table, with each one beginning at the far left. \) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
